PSI Explained: Process Chemistry
The information pertaining to the technology in the process must also include a description of any chemistry involved in the process. For processes which manufacture chemicals this is particularly important since the chemical reactions are the basis for the existence of the process. Process chemistry is trivial for processes in which a chemical is merely stored and then sold. Other processes, such as ammonia refrigeration, which utilize a chemical in a closed loop that changes state (liquid/vapor), typically use Process Chemistry to document the thermodynamic behavior of the chemical in the process.
Many of our ammonia refrigeration clients use the following paragraphs describing “How an Ammonia Refrigeration System Works” to document Process Chemistry:
A refrigeration system is used to reduce the temperature of a heat source. A mechanical refrigeration system is a closed system that cycles the refrigerant, ammonia, from liquid to gas and back again. As the refrigerant absorbs large amounts of heat from the source, the refrigerant changes from liquid to gas. At a different part of the system, the refrigerant releases heat as it is condensed back into a liquid. This system is referred to as having two sides: a low pressure or low side, where the refrigerant absorbs heat as it evaporates, and a high pressure or high side, where the refrigerant vapor releases heat as it is condensed back into a liquid. The system basically moves heat from the low side to the high side. Refrigeration is maintained by isolating the low side in some form of insulated enclosure where goods or processes can be chilled or frozen. The figure below displays a typical layout of a simple ammonia refrigeration system. The refrigeration system employed at Company XYZ is more complex and is detailed below.
Typical Ammonia Refrigeration System
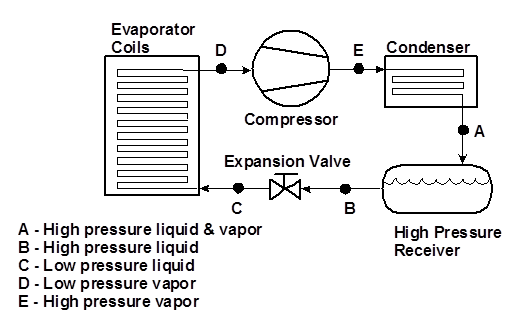
The low side of the refrigeration system typically uses one or more evaporators to remove heat from the space or products to be cooled. An evaporator contains a set of continuous coils through which the refrigerant flows. Due to the low pressures and corresponding low temperatures maintained in the evaporator, the liquid refrigerant quickly evaporates, absorbing a great deal of heat from the surrounding area. To increase the rate of heat removed, the evaporator uses an electric fan to move warm air over the coils of cold liquid ammonia.
As can be seen in the figure, the low and high sides of the refrigeration system are separated by the compressor and the expansion valve. The compressor is the driving force of the system. It pulls refrigerant vapor from the evaporator (low side) at a rate that will maintain the evaporator pressure required for the desired operating temperature. At the same time, it pressurizes the warm vapor and forces it into the condenser and the high side of the system. Refrigeration compressors on the market today are screw type, reciprocating, and centrifugal.
Expansion valves regulate the flow of liquid refrigerant from the high side to the low side by causing a large pressure drop. These valves are located between the receiver (high side) and the evaporator (low side). This valve, coupled with the capacities of the compressor, can help regulate the pressure of the evaporator thereby creating the cooling capabilities of the system.
The high side of the refrigeration system consists of a condenser and a receiver. Warm refrigerant vapor enters the condenser after it has been pressurized by the compressor. A condenser will typically use air and water to cool the warm ammonia as it passes through a series of tubes within the condenser. As the cooling medium of air and water draws the heat from this vapor, the refrigerant condenses into a liquid and flows to the receiver. The receiver serves as a holding tank for the liquid refrigerant being fed to the low side. It also serves to hold the bulk of the refrigerant in the system when it is not operating.

Leave a Reply