Properties of Refrigerants: Refrigerant Numbers
What do the various refrigerant numbers mean? Believe it or not, there is a method to the madness.
First of all, most refrigerants are “organic compounds,” which means they have one or more carbon-hydrogen or carbon-carbon bonds. The only series classified as “inorganic”, or lacking these bonds, is the 700-series, which includes ammonia, carbon dioxide, and water. 
Most of the organic compounds used as refrigerants are hydrocarbons, meaning they are composed of carbon and hydrogen. Common examples of hydrocarbons are methane, ethane, propane, and butane. 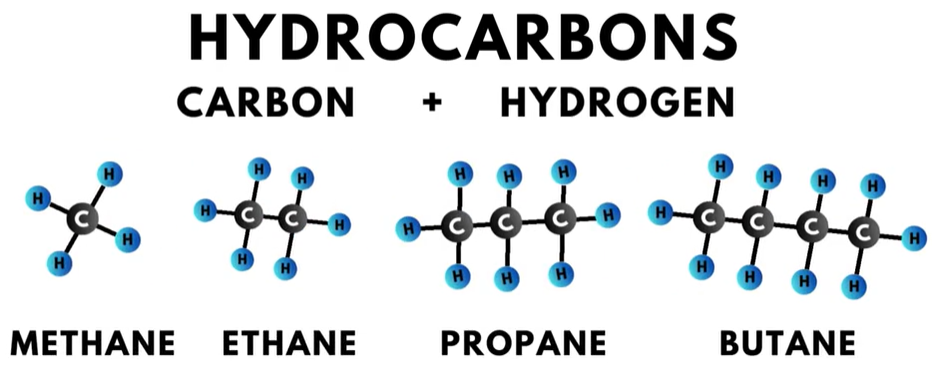
Hydrocarbons are highly flammable, so scientists in the 1920s discovered that by trading a halogen atom for one or more of the hydrogen atoms, the resulting compound would be “safer” to use. Halogens are the elements in the second column from the right on the periodic table. Chlorine and fluorine are common halogens that have been historically used in refrigerant compounds. These new refrigerants were named “halogenated hydrocarbons”. Over time, that name was shortened, and they became known as “halocarbons”, a term still used today.
Here is an overview of the nine series of refrigerants:
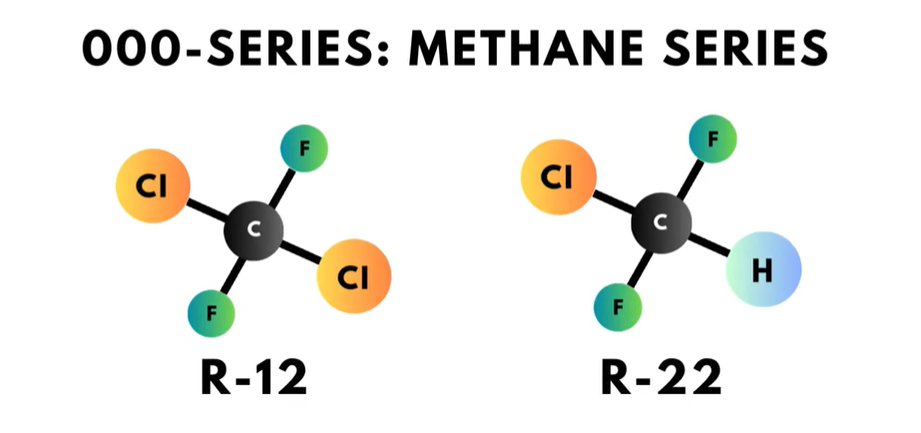
- The triple zero series is referred to as the “methane series”. Each compound in this series has a single carbon atom. The zero in the hundreds place of the refrigerant number in this series is omitted, so this series of refrigerants only has two digits. R-12 and R-22 are two of the refrigerants in this series.
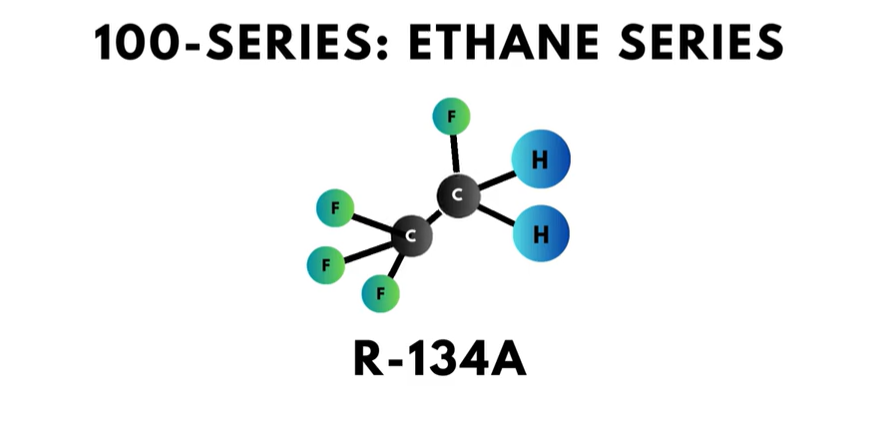
- The 100-series is the “ethane series,” and each compound has two carbon atoms. R-134a is a well-known refrigerant in this series.
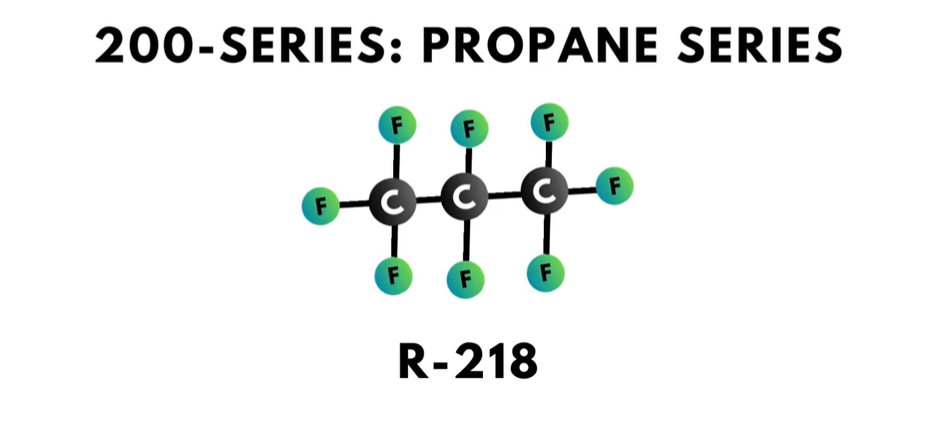
- The 200-series is the “propane series”. Each compound in this series has three carbon atoms.
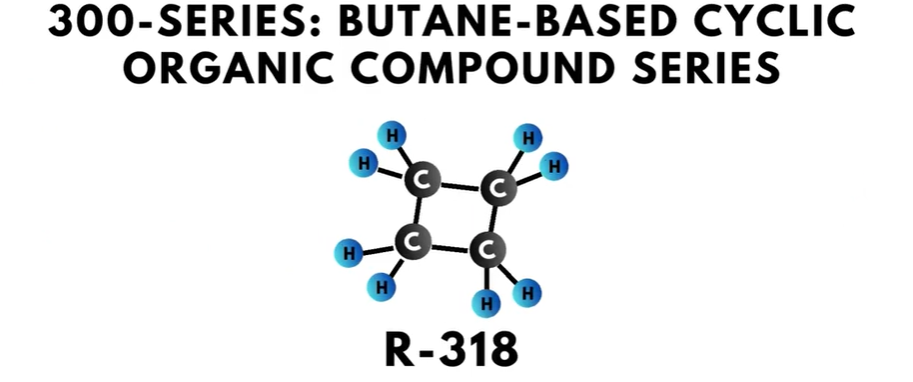
- The 300-series is the “butane-based cyclic organic compound series”. Compounds in this series have four carbon atoms.
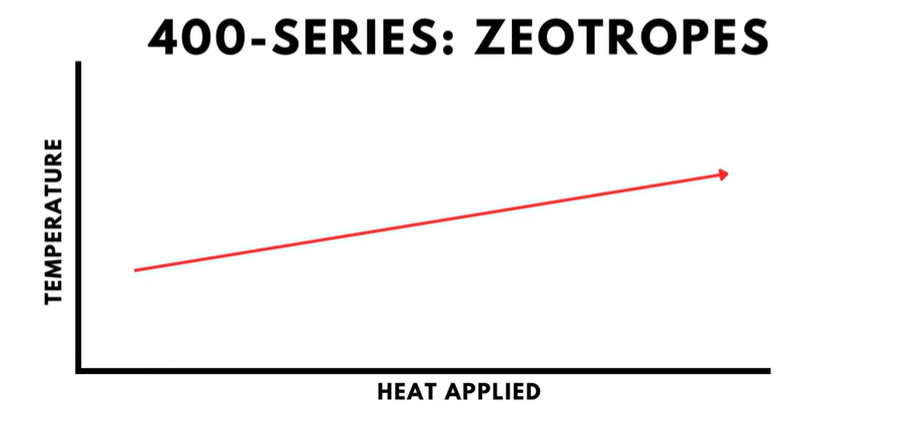
- The 400-series is reserved for refrigerant mixtures called “zeotropes”. Zeotropes tend to separate during boiling, which causes the refrigerant to have a temperature change, called a “glide”, during boiling.
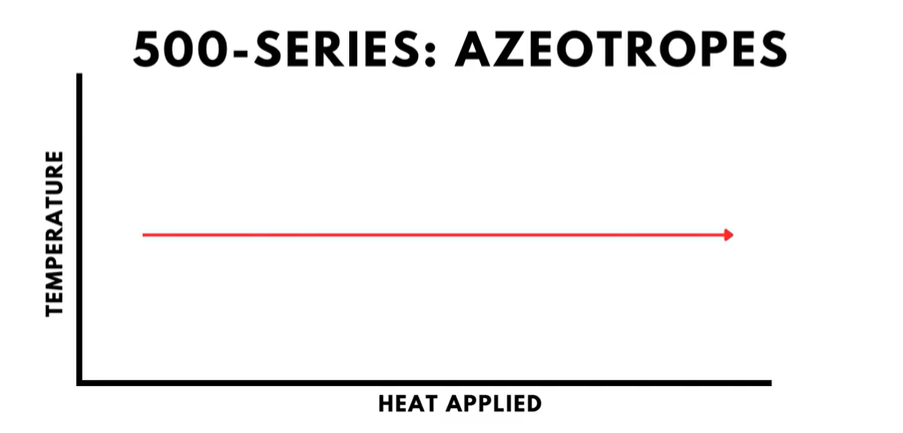
- The 500-series is for a different group of refrigerant mixtures called “azeotropes”. Azeotropes boil at a constant temperature and therefore behave similarly to pure refrigerants.
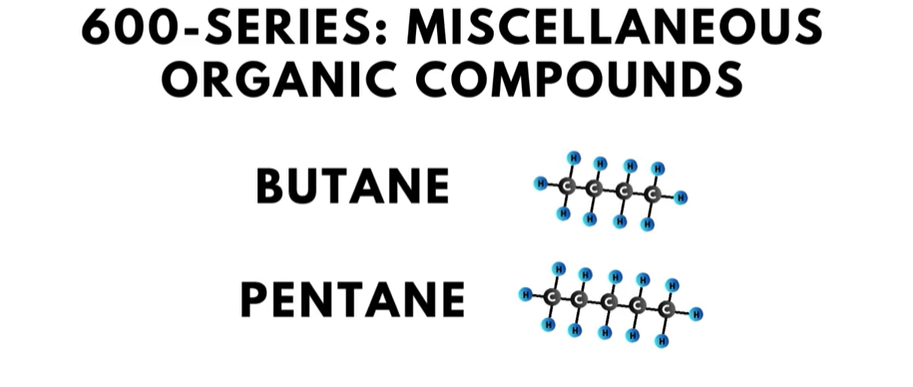
- The 600-series of refrigerants is for “miscellaneous organic compounds” such as butane and pentane.
- As mentioned earlier, the 700-series is for “inorganic compounds” such as ammonia, water, carbon dioxide, and sulfur dioxide.

- There is also a 1000-series of refrigerants that is reserved for refrigerants with “unsaturated organic compounds”. “Unsaturated” means that these organic compounds have one or more carbon atoms linked by a double bond.
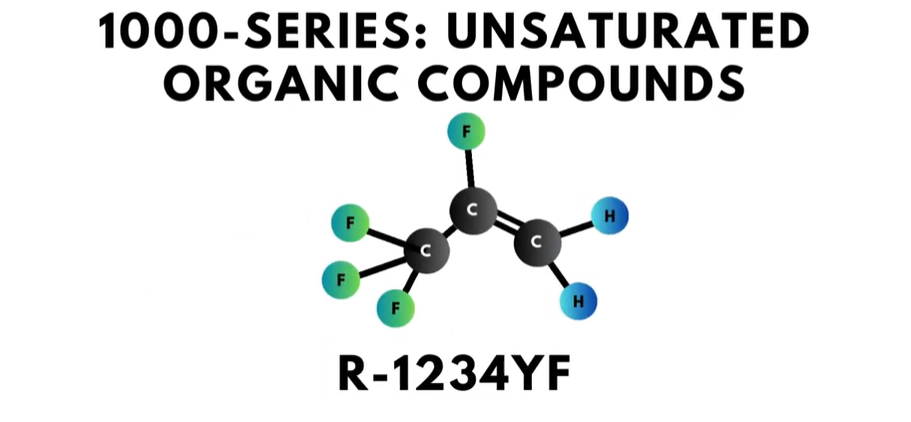
The first digit on the right, or in the “ones” place, is the number of fluorine atoms in the compound. For example, R-22 has two fluorine atoms.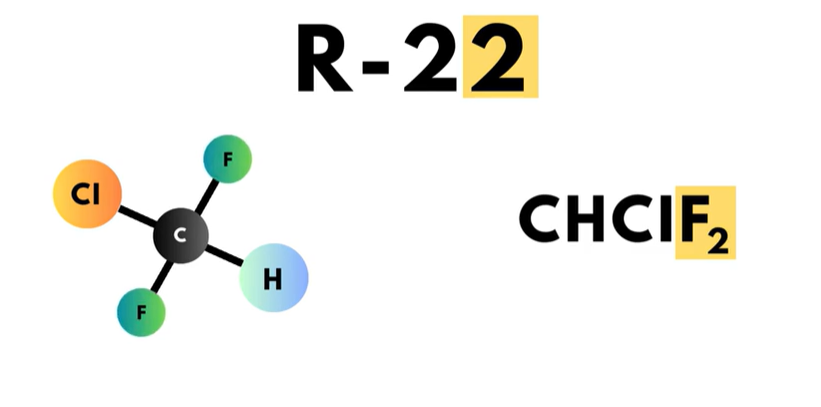
The second digit to the right, or in the “tens” place, is one more than the number of hydrogen atoms in the compound. Looking at R-12, there is a ‘1’ in the tens place, so that means that R-12 does not have any hydrogen atoms.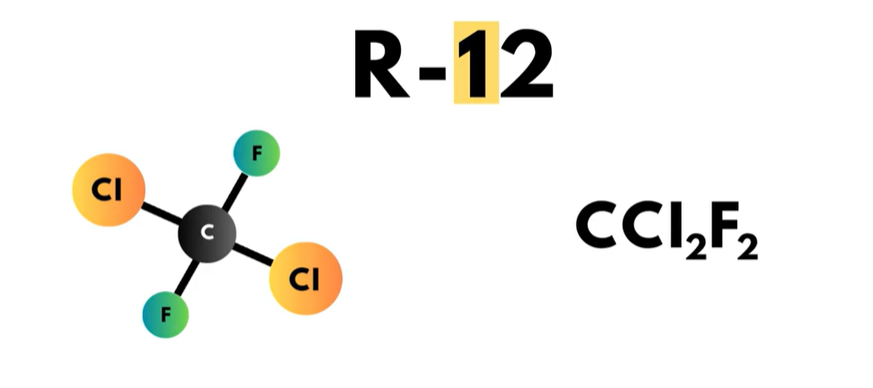
The third digit to the right, or in the “hundreds” place, is one less than the number of carbon atoms in the compound. Propane, which has the refrigerant number R-290, has a ‘2’ in the hundreds place, which means that the compound has three carbon atoms.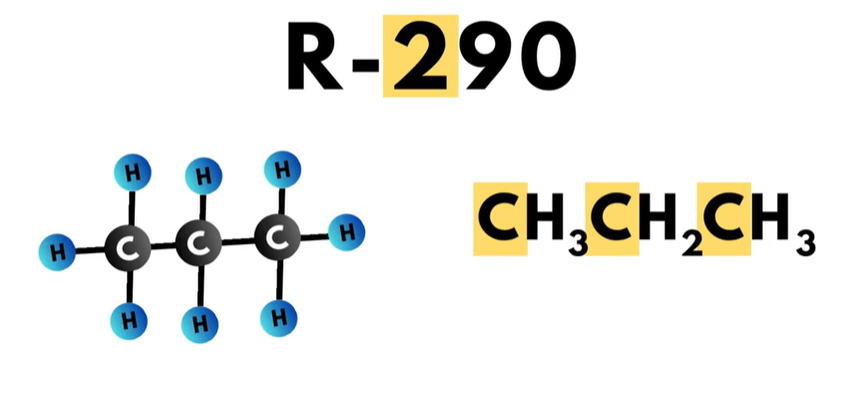
There is a fun trick called “the Rule of 90” that works as follows: adding 90 to the refrigerant number of any refrigerant below R-400 will result in the numeric value of the number of carbon, hydrogen, and fluorine atoms. Applying “the Rule of 90” to R-22 works like this…
22 plus 90 equals 112. This means that R-22 has one carbon, one hydrogen, and two fluorine atoms.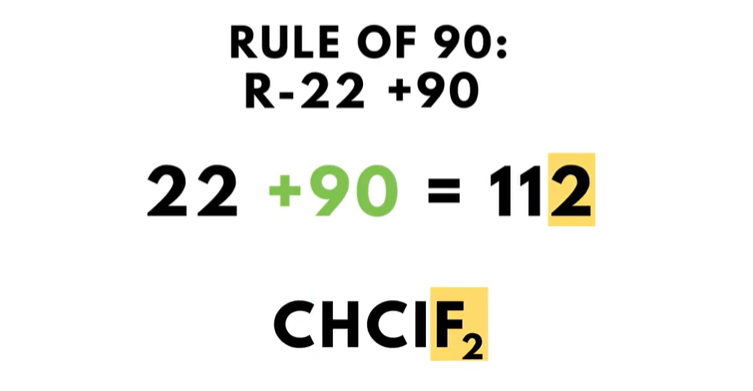
Remember, this does not work for the 400 through 1000 series refrigerants.
Example:
Apply the “rule of 90” to R-134a.
134 + 90 = 224
This refrigerant has 2 carbon, 2 hydrogen, and 4 fluorine atoms. The chemical formula for R-134a is CH2FCF3
The 700-series is for inorganic compounds such as ammonia, carbon dioxide, and water. In this series, the ‘7’ in the hundreds place simply identifies the refrigerant as an inorganic compound. 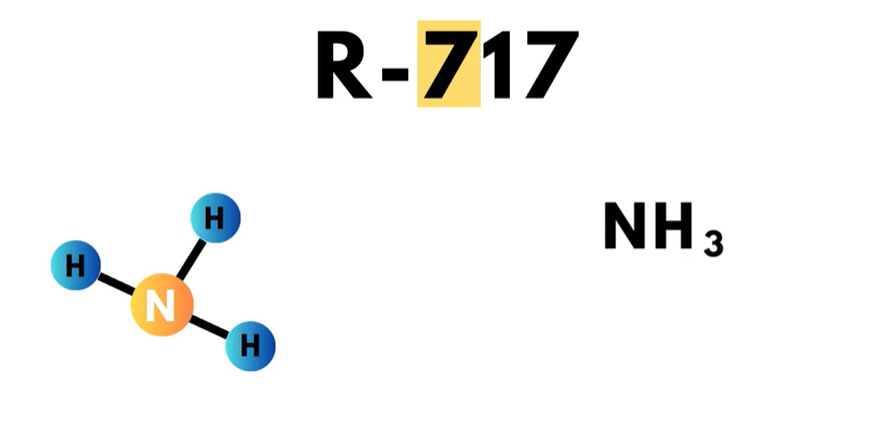 The final two digits are the molecular weight of the compound. For example, ammonia is a refrigerant number R-717. The first ‘7’ indicates it is an inorganic compound, meaning it has no carbon-hydrogen bond. The ‘17’ is the molecular weight of ammonia, which has the chemical compound NH3. Nitrogen and hydrogen have molecular weights of 14 and 1, respectively. And it is easily seen that 14 + 1 + 1 + 1 = 17. Hence, the refrigerant number of R-717.
The final two digits are the molecular weight of the compound. For example, ammonia is a refrigerant number R-717. The first ‘7’ indicates it is an inorganic compound, meaning it has no carbon-hydrogen bond. The ‘17’ is the molecular weight of ammonia, which has the chemical compound NH3. Nitrogen and hydrogen have molecular weights of 14 and 1, respectively. And it is easily seen that 14 + 1 + 1 + 1 = 17. Hence, the refrigerant number of R-717.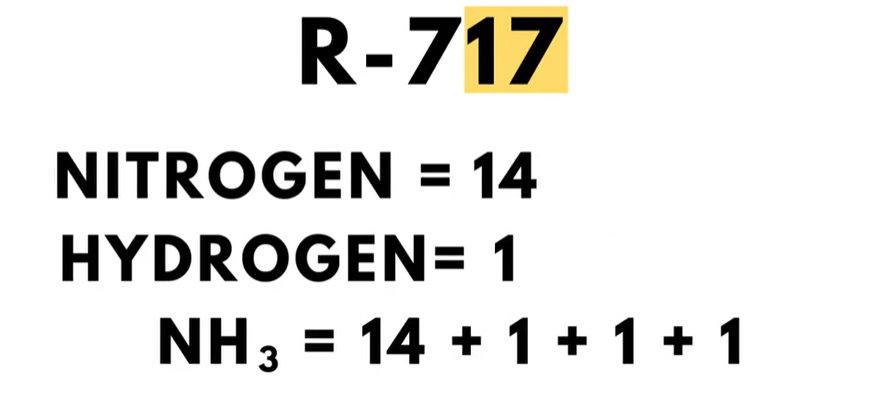

Leave a Reply