Fundamentals of Refrigeration: States of Matter

Simply defined, “matter” is anything that takes up space and can be weighed. All physical things are made of matter since physical things have both volume and mass. Technically, there are five states of matter:
- Solid
- Liquid
- Gas
- Plasma
- Bose-Einstein condensate
Items four and five on that list are not relevant to the study of refrigeration fundamentals, so the focus will be on the first three states of matter listed.

In “solid” materials, molecules are tightly packed together.
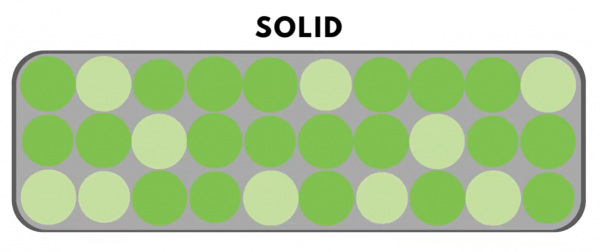
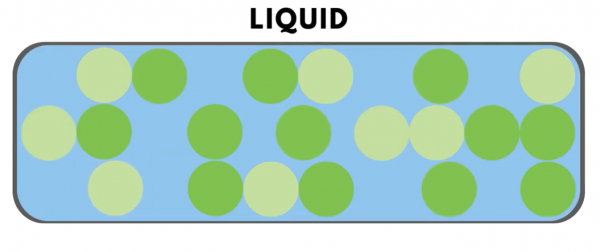
In “liquids,” the particles can move more freely and slide past each other.
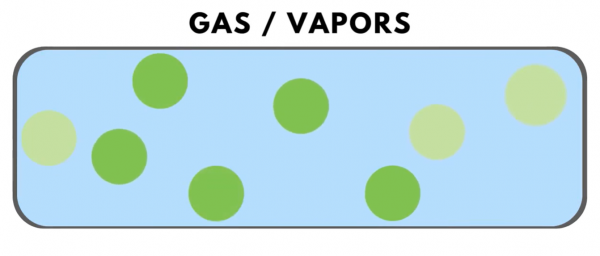
In “gases” or “vapors,” the molecules are moving very fast and have even greater spacing from each other.
The term “change of state” refers to the process of a substance changing from one state of matter into another. A synonymous term is “change of phase”.
“Condensation” refers to the process of a vapor becoming a liquid.

“Boiling” or “evaporation” refers to the opposite process when a liquid turns into a vapor.

“Melting” is the phase change from solid to liquid, and “fusion” is the reverse process whereby a liquid becomes a solid.

Less common terms are “deposition” and “sublimation”. “Deposition” is the transition of a substance from a vapor directly to a solid. The formation of snow is an example of deposition. “Sublimation” is the reverse process where a solid substance transforms into a gas. An example is dry ice, which sublimates to become carbon dioxide gas.

Leave a Reply