Fundamentals of Refrigeration: Pressure/Temperature Relationship

When a substance changes state from a liquid to a vapor, the process occurs at a constant temperature. When this occurs in a refrigeration system, it is said that the refrigerant is “saturated”. For water, experiments have proven that saturation, or boiling, occurs at 212ºF at 14.7 psia, which is atmospheric pressure.

However, if someone were to travel to Denver, Colorado, which has an altitude of a mile, they would find that water boils at 203ºF. What caused this change?
As the altitude changes, the atmospheric pressure changes. At an altitude of one mile, the atmospheric pressure is 12.2 psia, which causes a change in the saturation temperature.
This is important because it teaches us that the saturation temperature is dependent on the pressure. Scientists have performed experiments to determine the saturation temperatures of refrigerants at various pressures and developed useful tables that engineers can use when selecting refrigerants. We call these the “saturation tables”. Additionally, a German scientist named Richard Mollier plotted the values on diagrams that are named after him, “Mollier Diagrams”.
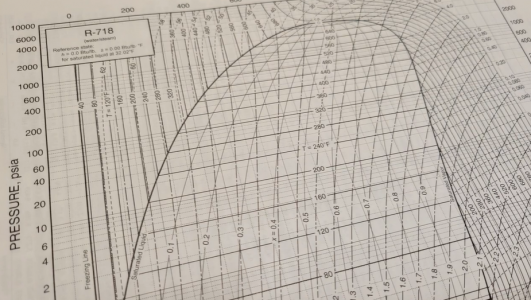
The relationship between pressure and temperature is extremely helpful because it means that when the pressure of a saturated refrigerant is measured, the temperature will also be known, and vice versa
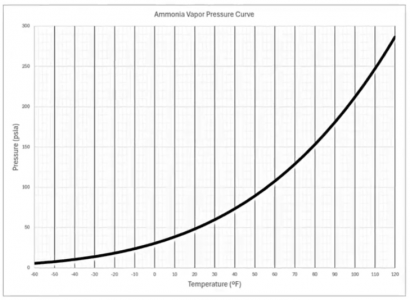
The graph pictured below depicts the saturation points for ammonia. The line communicates to us the temperature and corresponding pressure when ammonia is saturated. If ammonia in a refrigeration system does not fall on this line, it means that the ammonia is subcooled or superheated.

Leave a Reply