Properties of Refrigerants: Halocarbon Refrigerants
Halocarbons are man-made compounds. The term halocarbon is shorthand for “halogenated hydrocarbon”. A halocarbon is a hydrocarbon compound that has replaced at least one hydrogen atom with a halogen atom.
Halocarbons have been used for many purposes, such as Solvents, Fire Suppression, and Refrigeration.
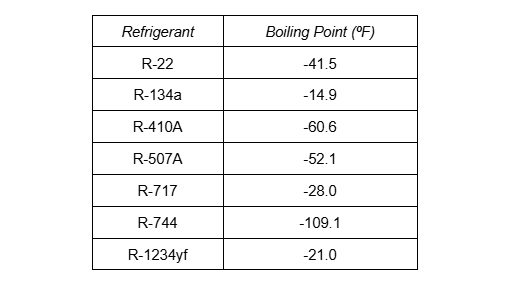
Halocarbons boil at various temperatures. Based on the chemical formula, each compound has a different boiling point at atmospheric pressure. The table on the screen displays the boiling point for some common halocarbons, natural refrigerants, and unsaturated hydrofluorocarbons, or HFOs.
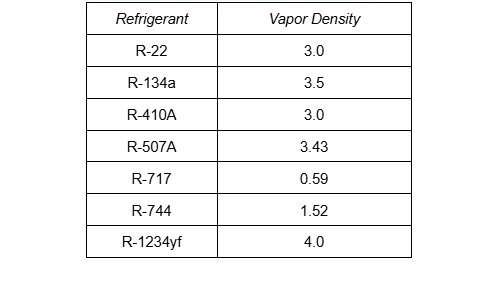
Halocarbons are always heavier than air. This is not ideal, since it means that during a leak, halocarbons will tend to accumulate near the floor, replacing the oxygen humans need to survive. As you can see, all of the halocarbons are at least three times heavier than air.
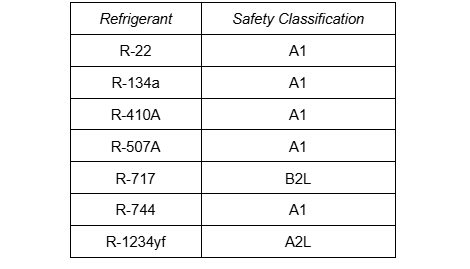
Most common halocarbon refrigerants are non-flammable and non-toxic, which has been a primary advantage over ammonia throughout the years. ‘Class A’ is assigned to refrigerants with low toxicity and ‘Class 1’ is assigned to non-flammable refrigerants. This table depicts the safety group for several common refrigerants. Notice that all halocarbons shown have an ‘A1’ classification. Even with a classification of ‘A1’, a slight fire hazard can exist with halocarbons. This is because halocarbons are soluble and miscible with oil.
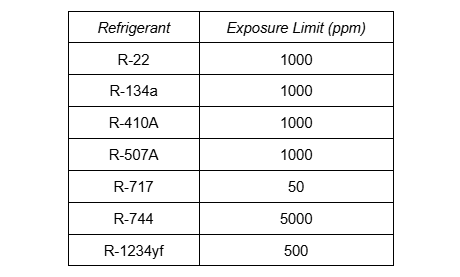
Exposure limits, such as NIOSH’s recommended exposure limit, REL, or OSHA’s permissible exposure limit, PEL, are the limits of exposure to a chemical substance that are considered safe without respiratory protection. Due to their non-toxic nature, most halocarbons have much higher exposure limits compared to ammonia. The table here displays the exposure limits obtained from the safety data sheets of several common refrigerants.
Halocarbons are readily available from refrigerant suppliers. Halocarbons have historically been more expensive than natural refrigerants, such as ammonia. In recent years, the price of halocarbon refrigerants has skyrocketed due to restrictions on manufacturing and importing halocarbons.

Halocarbons are considered odorless, so leak detection is more difficult compared to ammonia. Technicians, therefore, must utilize an electronic leak detector or halide torch to find a leak.
In general, halocarbons do not react with other chemicals and unlike ammonia, halocarbons are compatible with copper. As a result, copper is widely used as the material of choice for piping and heat transfer surfaces in halocarbon systems.

Halocarbons are heavily regulated because of their ozone depletion and global warming potential. The United States signed on to the Montreal Protocol in 1987, which led the way to the Clean Air Act in 1990. The Clean Air Act has given the EPA authority to restrict the use and phase out refrigerants deemed harmful to the environment.


Leave a Reply