Fundamentals of Refrigeration: Saturation, Subcooling, and Superheating

“Saturation” is a term used to describe a substance that is at its boiling point. Water, for example, is said to be saturated when its temperature is 212ºF at sea level. The 212ºF water may be a liquid, a vapor, or a mixture of the two. When a substance is saturated, heat added or removed from the substance will be in the form of latent heat and will not result in a temperature change.

In a refrigeration system, whenever a vessel or heat exchanger contains both liquid and vapor, the refrigerant is saturated. When a substance is saturated, the saturated tables can be used to determine the properties of the substance if the temperature or pressure is known.
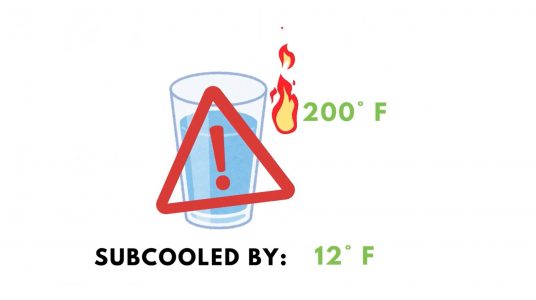
A substance is said to be “subcooled” when it is cooled below its boiling point, or saturation temperature. Don’t let the word “cool” deceive you into thinking this is referring to a “cold temperature”. When water is at 200ºF, it is subcooled by 12ºF. The water is dangerously hot, but subcooled, nonetheless. When a substance is subcooled, any heat added or removed from the substance will be in the form of sensible heat and will result in a temperature change.
The term “superheated” is used to describe vapors that are heated above their saturation point. 230ºF steam has 18ºF of superheat. When heat is added or removed from a superheated vapor, the temperature of the vapor will change, meaning the heat is in the form of sensible heat. Refrigerants exiting a compressor will always be superheated.

Leave a Reply