Fundamentals of Refrigeration: Heat Energy Profile of Water

To reinforce what is known about the states of matter, it can be helpful to study the heat energy profile of water. The graph below shows heat, measured in BTUs, applied to a pound of water on the horizontal axis and the temperature change of the water on the vertical axis. The values displayed are for standard atmospheric pressure, or 14.7 psia.
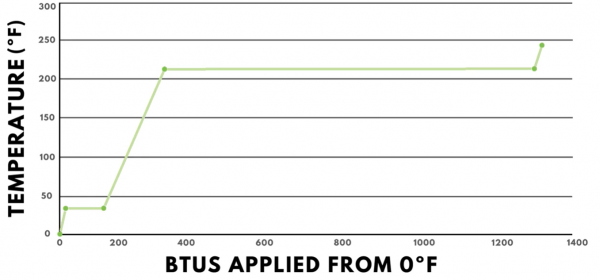
Starting at 0ºF, water is in its solid state of matter, which is called ice. Every BTU added causes us to move up and to the right. This is because the heat added is “sensible heat,” which means it causes the temperature to change. The specific heat of ice is 0.5 BTUs per pound degree Fahrenheit, so adding one BTU results in a two-degree Fahrenheit temperature change.
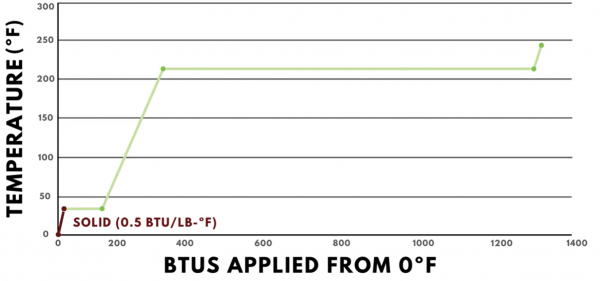
As more heat is added, the graph continues on a straight line until it reaches 32ºF. 32ºF is the melting point of ice, so once it reaches this temperature, all of the heat added contributes toward changing the state of matter from solid to liquid, or from ice to water. The latent heat of fusion of water is 144 BTUs per pound, so the line is perfectly horizontal as it moves 144 BTUs to the right.
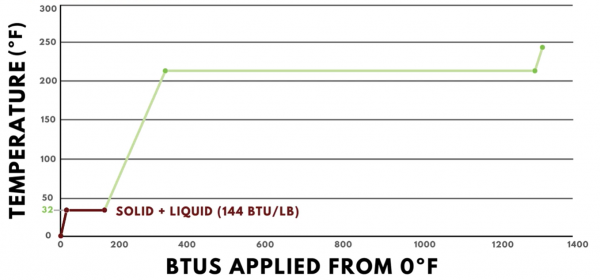
Once the ice has fully transformed into water, additional heat added will start changing the temperature again. However, the specific heat of water is 1 BTU per pound degree Fahrenheit, so the slope of the line in this region is less steep than it was in the sensible heat region for ice. For every BTU of heat added, the temperature of one pound of water increases by one degree. It continues along this path until it reaches the boiling point of water, which is 212ºF.
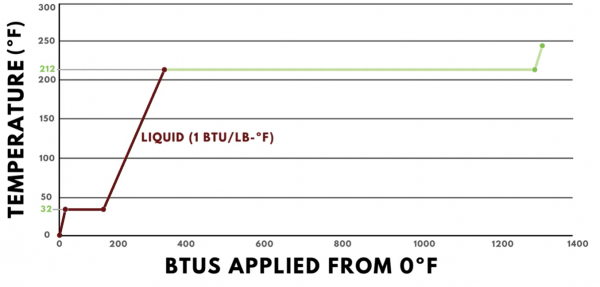
At 212ºF, the phase change to steam begins, which is the vapor form of water. A significant amount of heat is required to change one pound of water into steam, 970 BTUs to be exact, which is called the latent heat of vaporization. Because a change of state is occurring, all of the heat added is in the form of latent heat, and there will be no change in the temperature.
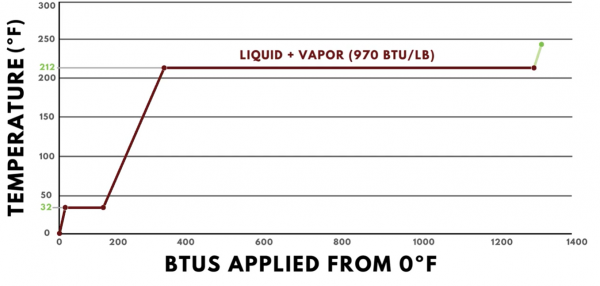
Once the pound of water has become fully steam, additional heat will once again result in a temperature change. The specific heat of steam is 0.5 BTUs per pound degree Fahrenheit, so each BTU added will change our pound of steam by 2 degrees Fahrenheit.
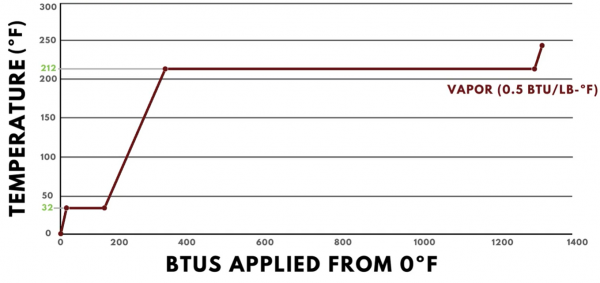
The heat energy profile of water illustrates many things that are important to refrigeration.
First, when heat is added to water in a single state, it will result in a temperature change. Since this heat can be measured with a thermometer, it is called “sensible heat”.
When heat is added results in a change of state, and it is referred to as “latent heat” because there is no change in temperature.
Finally, the heat required to change phases is significantly greater than the heat required to change temperature within a single phase. More specifically, the heat required to change state from liquid to vapor is substantial. This concept is crucial to understanding refrigeration. Even though this is the heat energy profile for water, other refrigerants follow a similar profile, so the lessons learned here can be applied.
Refrigerants are selected by their readiness to absorb and reject heat. A good refrigerant can accept a tremendous amount of heat in the transition from liquid to vapor. Therefore, one of the qualities an engineer considers when selecting a refrigerant is the latent heat of vaporization. The larger the value, the more effective the refrigerant will be.

Leave a Reply